Resources
Create a New CITI Program Account via UCLA SSO
Link an Existing CITI Program Account via UCLA SSO
NEW: CITI Webinars are now available with UCLA’s "All-Access Pass"
For questions contact:
Tanya Rosales Martinez
tanya.martinez@research.ucla.edu
(310) 825-4994
For general training comments, email OHRPPEQI@research.ucla.edu
How do I obtain proof that I've completed GCP training for ICH version 3?
Some sponsors require proof of a specific GCP training for the current ICH version.
The biomedical GCP course available through UCLA's CITI account was automatically updated to reflect ICH version 3 on 10/15/2025.
If you complete all modules for the course after 10/15/25, you can download a certificate (available in your account in CITI) with a statement indicating ICH version 3 on it.
If you completed some or all modules for the course before 10/15/25, you will need to re-take those modules in order to obtain a certificate with the ICH version 3 statement on it.
In addition, we have added to UCLA's CITI account a short course titled "ICH E6(R3): An Introduction." This course describes the changes to section E6 (GCP) from ICH revision 2 to ICH revision 3. This is an optional course, but available to you if you want it or if your study Sponsor requires it.
What is CITI Training?
The Collaborative Institutional Training Initiative (CITI) Program is a leading provider of research education content. Its web-based training materials serve millions of learners at academic institutions, government agencies, and commercial organizations in the U.S. and around the world. UCLA has chosen to subscribe to the CITI Program for multiple research training requirements, including for human subjects research.
Am I required to complete Human Subjects Protection training? When?
All UCLA Key Personnel involved with human subjects research must have a current and complete UCLA Human Subjects Research certification or UCLA Good Clinical Practice (GCP) certification via CITI prior to inclusion in the IRB application (either via initial submission, amendment, or through use of the "edit study personnel" function):
- a new study on which they are named
- an amendment application adding them to a previously approved study
- a continuing review application for a study on which they are named
The BruinIRB system will conduct automatic checks to ensure all key personnel have current human subjects training. If any key personnel have missing or expired training, or if the personnel’s CITI account is not linked to their UCLA Single Sign On (SSO), the system will block submission of the application.
- Please view the applicable guidance document below for details about how to link your existing CITI account or how to create a new linked account:
Principal Investigators are responsible for assuring that all Key Personnel on the study have completed the UCLA CITI training requirements.
- Key personnel on any study may choose to complete GCP training in lieu of human subjects research training.
- Key personnel on NIH- funded clinical trials must complete GCP certification (either in addition to or instead of Human Subjects Research certification).
If UCLA is relying on another institution’s IRB to review your study, then UCLA is usually required to certify to that IRB that all UCLA Key Personnel have completed UCLA’s CITI requirements.
If UCLA is the reviewing IRB for your collaborating sites, then those sites are usually required to certify to UCLA that their Key Personnel have met those sites’ own training requirements.
CITI training completed at another institution may or may not transfer for credit at UCLA, depending on how recently it was completed, and the type of course(s) completed at the other institution. To determine if previously completed coursework in CITI will transfer, affiliate with UCLA in your existing CITI account, link your existing account to your UCLA single sign on (SSO), then follow the prompts to “Add a course”. At the end of that course selection process, any modules recently completed for another institution that overlap with the UCLA CITI course(s) enrolled will automatically be credited to your UCLA CITI course(s) (and will not have to be completed again).
Key Personnel is defined as the Principal Investigator, other investigators and research personnel who are directly involved in conducting research with study participants or who are directly involved with handling private information related to study participants during the course of a research project. Key Personnel also include Faculty Sponsors who oversee student Principal Investigators in their conduct of human subjects research.
Am I required to take CITI HIPAA Research Training?
CITI HIPAA is required for all UCLA researchers involved in research involving human participants that creates, uses, and/or discloses Protected Health Information (PHI). This includes research that involves the review of medical records or the creation of new medical records or the addition of information to existing medical records.
If you completed the UCLA HIPAA Research training certification course that was offered PRIOR TO 2009 this meets UCLA’s HIPAA Research training requirement and you do not need to complete the UCLA HIPAA course on CITI.
There is no refresher course required for UCLA HIPAA research training.
For additional information on HIPAA, please see the UCLA OHRPP guidance on HIPAA Research Guidelines and Information.
How do I access CITI training?
Anyone who is required to take our UCLA CITI courses must first create an account within the CITI website or affiliate themselves with UCLA if they already have an existing CITI account. All new CITI accounts must be created using your UCLA single sign-on ID. If you do not have a UCLA single sign-on ID, you may create one on the UCLA Identity and Accounts Manager website.
Please view the applicable guidance document below for details about how to link your existing CITI account or how to create a new linked account:
Please note that linking your CITI account to your UCLA single sign-on ID will not prevent access after leaving UCLA.
Do I need to renew Human Subjects Protection Training?
Key Personnel must renew their Human Subjects Research or GCP training via CITI every 3 years. Depending upon the course selected to satisfy this requirement (GCP or HS), you will either complete the preselected refresher modules, or the 4 elective modules of your choice.
You will receive an email notice from CITI 180 days, then 90 days, then 30 days prior to the expiration date for your course, but you may also log on to the CITI website to see when you are due to take your Refresher Course. You may complete the Refresher Course up to 180 days (that is, six months) prior to the expiration date of your current CITI training.
- For those with existing Human Subjects (HS) certification, at the time of renewal, CITI will automatically enroll you in the HS refresher course. If you would prefer to complete GCP training instead of the HS refresher course, you may do so. Inside your UCLA CITI account, click the "Add a course" button to add a GCP course and click the "Remove a course" button to remove the HS refresher course you no longer wish to take
How can I verify completed training for myself and key personnel on my studies?
There are several places to verify that you and/or key personnel on a study have completed training.
(1) Key Personnel on a Study:
Within the BruinIRB application, CITI training will appear on the Local Study Team Members section for all personnel as they are added to the application:

(2) Personal Training Documentation:
Within the Profile section of BruinIRB, you can confirm your training dates and correct linking with BruinIRB:
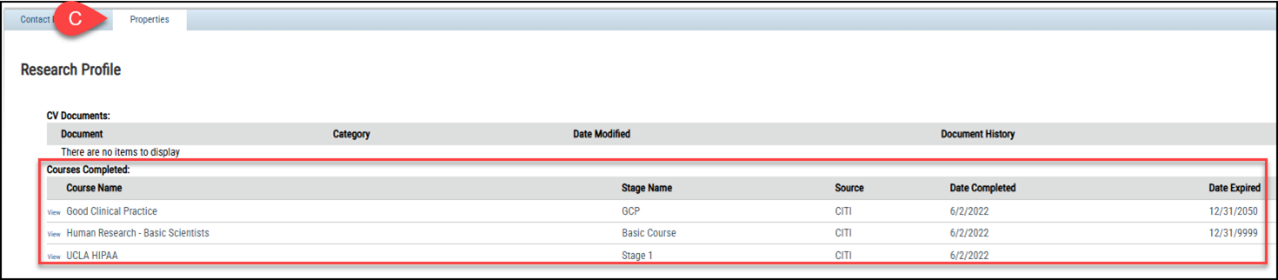
a. Click on your name in the upper right-hand corner of the screen.
b. Click on My Profile.

c. Click on the Properties tab. Any training completed through CITI will be listed here with the date completed and expiration date, if applicable.
(3) CITI Training Lookup Tool:
Search for completed trainings by last name, first name, department, and/or University ID number. Please note that this tool requires logging into the ORA Portal using your UCLA Logon ID.

BruinIRB performs automatic training checks. If you have completed CITI training and your name appears in the CITI Lookup Tool, but not the IRB application, you will need to link your UCLA Single Sign-On (SSO) information to your CITI account. Please see UCLA Single Sign-On (SSO) Integration for CITI Program Training for detailed information on linking your account.
Please Note: Completion of CITI training does not automatically create an IRB account for you. If you need access to view or edit studies, please see the Electronic Submission Systems page.
Where can I get more information?
For additional information on the CITI training requirements and how to access the training refer to Frequently Asked Questions.
